|
#1
| |||
| |||
| |
|
#2
| |||
| |||
|
My sister needs syllabus of 3rd Semester of M.SC Chemistry Course (Inorganic) of Dr. Babasaheb Ambedkar Marathwada University (BAMU). So can anyone provide syllabus of 3rd Semester of M.SC Chemistry (Inorganic) Course of Dr. Babasaheb Ambedkar Marathwada University (BAMU)?
|
|
#3
| |||
| |||
|
Here I am providing complete syllabus of 3rd Semester of M.SC Chemistry Course (Inorganic) of Dr. Babasaheb Ambedkar Marathwada University (BAMU): Dr. Babasaheb Ambedkar Marathwada University (BAMU) M.SC Chemistry 3rd Semester (Inorganic) CHESC-301 Structural elucidation by spectral methods CHECI- 302 Bioinorganic and supramolecular chemistry CHECI- 303 Applied inorganic chemistry CHEEI -304 Chemistry of materials OR CHEEI -305 Environmental chemistry OR CHEEI- 306 Solid state chemistry CHELI- 307 Laboratory course CHELI- 308 Laboratory course CHELI- 309 Laboratory course CHESC-301: Structural elucidation by spectral method UNIT-I: Nuclear Magnetic Resonance Spectroscopy (1H NMR) [12hrs] Elementary ideas (Recapitulation); Spin-spin couplings, Different types of couplings, factors affecting on coupling constants, Karplus equation, Spin systems (AB, AX, ABX, AMX), Rate processes, spin decoupling, shift reagents, Nuclear Overhauser effect (NOE), INEPT and INADEQUATE. UNIT-II: 13CNuclear Magnetic Resonance Spectroscopy [12hrs] Elementary ideas, instrumental problems, chemical shifts (aliphatic, olefinic, alkyne, aromatic, heteroaromatic and carbonyl carbons); Effect of substituents on chemical shifts. UNIT-III: Mass Spectroscopy [12hrs] Introduction, ion production (EI, CI, FD and FAB), ion analysis, ion abundance, factors affecting on fragmentation, fragmentation of different functional groups, molecular ion peak, isotopic peaks, metastable peak, Nitrogen rule, McLafferty rearrangement, Retro-Diels-Alder reaction. UNIT-IV [12hrs] Problems based on joint applications of UV, IR, 1H NMR, 13C NMR and Mass spectroscopy. UNIT-V [12hrs] Mossbauer spectroscopy: Principle, factors affecting the line position and shape, isomer effect and Quadrupole splitting iron salt like compounds, complexes, carbonyl compounds (temperature dependence of isomer shift and Quadrupole splitting in simple compound and coordination, polynuclear complexes), Numericals. Electron Spin Resonance Spectroscopy: Introduction, principle of ESR spectroscopy, presentation of spectrum, hyperfine splitting in various structures, hyperfine splitting diagram of representative examples, factors affecting the magnitude of ‘g’ values, Zero field splitting, Kramer’s degeneracy, Anisotropy in the hyperfine coupling constant, electron delocalization, instrumentation and applications. CHECI- 302: Bioinorganic and supramolecular chemistry Unit- I: Metalloenzymes: [12 hrs] Structural and functional relationships and mechanisms of enzymatic reaction in the following metalloenzymes. Zinc Enzymes: carbonic anhydrase and carboxypeptidases, Copper Enzymes: super oxide dismutases Iron Enzymes: catalase and peroxidases, Molybdenum Enzymes: nitrogenase and xanthanine oxidase,Coenzyme Vitamin: B-12 Unit-II: Metal Nucleic Acid Interactions: [12 hrs.] Introduction, nucleic acid structures, structures and binding sites in nitrogen bases, phosphates and sugar base. Coordination complexes of nucleic acids and their bases with metal ions. Hydrogen bonding, redox reaction and hydrolytic reaction of nucleic acids mechanisms of these reactions, natures role, pharmaceutical role, catalytic role Unit-III: Supramolecular chemistry [12 hrs] Basic concepts and principles of supramolecular chemistry, Host-Guest interactions, Molecular Recognition,spherical recognition, anionic receptors, porphyrin-sapphyrin systems, organometallic receptors, tetrahedral multiple and neutral molecular cleft recognition, enzyme models, molecular receptors, design and synthesis. Unit-IV: Supramolecular reactions and catalysis [12hrs] a. Catalysis by anion, cation and metal receptor molecules and co catalysis. b. Molecular and supramolecular devices : likes Photonic, Electronic and ionic devices.. Unit-V: Storage of metals and transport across the membrane [12hrs] The fluid mosaic model of membrane, types of transport and their mechanism, Transport and storage of alkali and alkaline earth metals, Na –K pump, calcium pump. Gibbs-Donnan equilibrium, Iron transport proteins and compounds. Download the attachment for syllabus: Dr. Babasaheb Ambedkar Marathwada University (BAMU) M.SC Chemistry 3rd Semester (Inorganic) 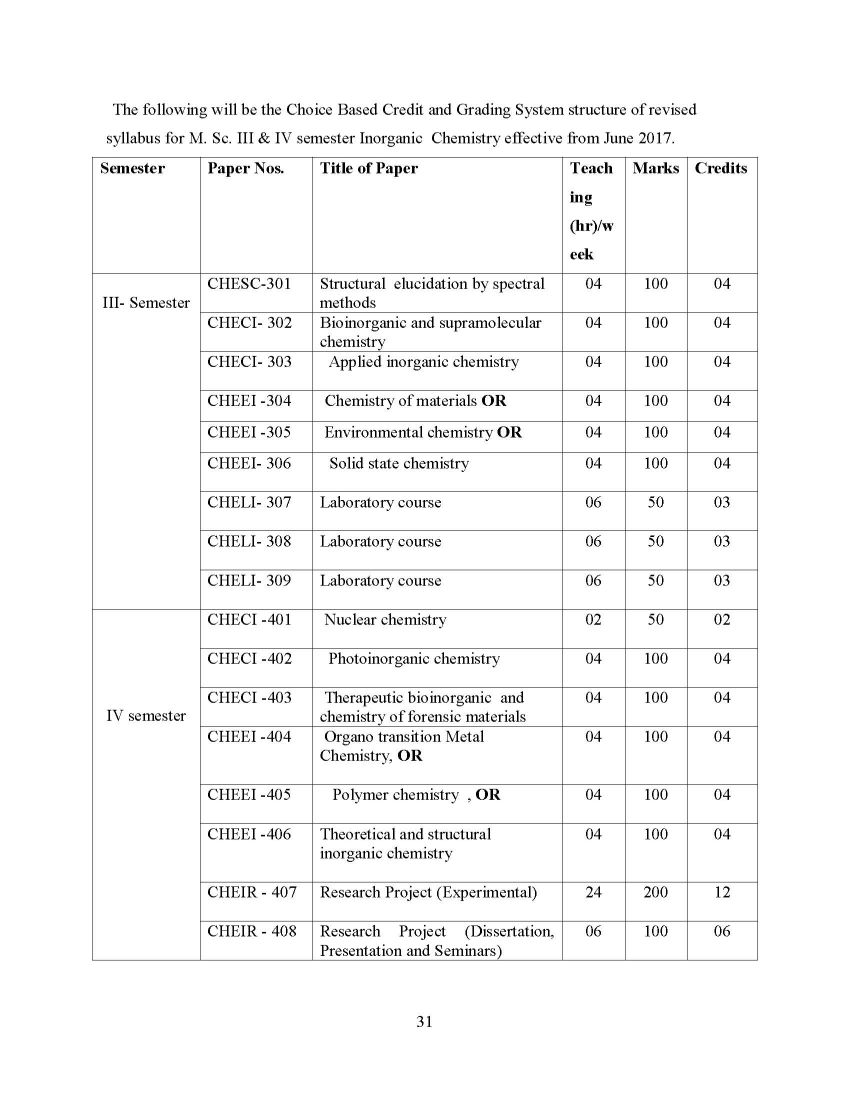    |
 |
| |